Immunization Storage and Handling
Cold chain refers to all the materials, equipment, and procedures used to keep immunizations in the required temperature range from the time they are manufactured until administration. The required temperature range for most immunizations is between +2oC and +8oC.
Maintaining cold chain is critical to immunization integrity. Immunizations are sensitive biological substances and failure to adhere to cold chain requirements may reduce their potency, resulting in lack of protection against vaccine preventable diseases and/or increased local reactions after administration. This loss of potency is cumulative, permanent and irreversible.
New Providers, Relocating, or Getting a New Immunization Refrigerator
Are you a new healthcare provider, organization or agency considering administering publicly funded immunizations?
Are you relocating your practice and need to move your immunization refrigerator?
Do you need to transfer immunizations from an existing vaccine immunization refrigerator to a new one?
Read through the information on this webpage and call the Health Unit (519-663-5317) to speak with a Public Health Nurse on the Vaccine Preventable Disease Team for support.
Cold Chain Inspections
Public health units in Ontario are required to perform annual inspections of all premises that store publicly funded immunizations.
What can you expect during your inspection?
During your inspection, a public health nurse from the Health Unit's Vaccine Preventable Disease Team will visit your office to:
- ensure the proper management of immunizations,
- review contingency planning,
- provide education and resources, and
- answer any questions your staff may have.
Immunization Refrigerators and Organization
Healthcare providers storing publicly funded immunizations are required to have a dedicated refrigerator used for the storage of immunizations only.
Types of refrigerators
The chart below provides a comparison of the types of refrigerators and some general guidelines.
| Purpose-built immunizations refrigerators | Domestic (kitchen) and bar refrigerators |
|
|
|
Guidelines for all immunization refrigerators |
|
|
|
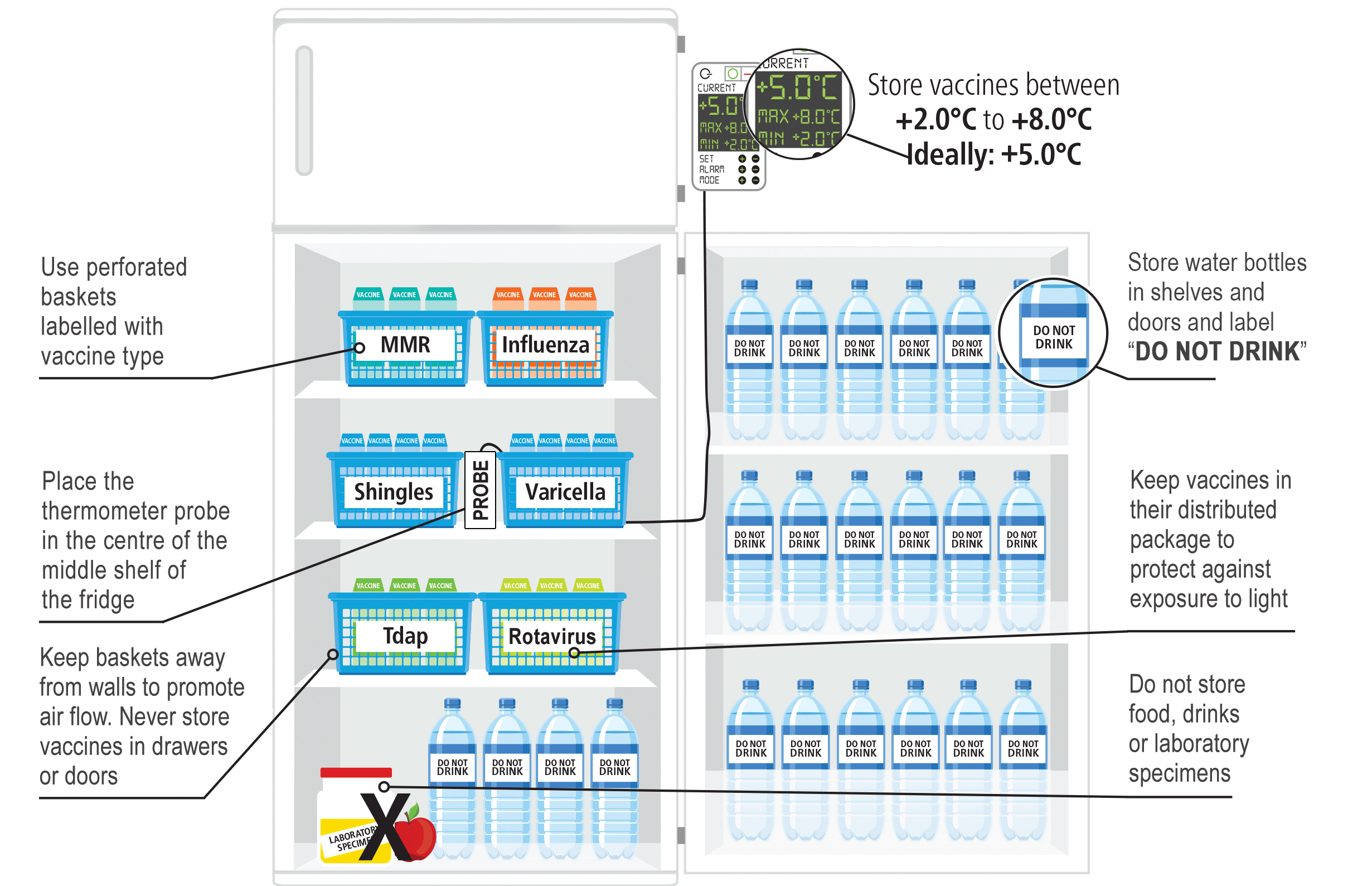
Organizing the refrigerator
Maintaining a well-organized immunization refrigerator plays an important role in:
- preserving immunization integrity (e.g. allowing ample air circulation, protecting immunizations from light),
- keeping temperatures within the recommended range (e.g. minimizes how long the refrigerator door is open), and
- preventing errors (e.g. administering an expired or wrong immunizations).
| Guidance for refrigerator organization | |
|
|
To prevent overcrowding and minimize waste in the evident of a cold chain incident, no more than a one month supply of immunizations should be stored in your vaccine refrigerator.
Temperature Monitoring
Each immunization refrigerator and insulated cooler used for immunization storage and transport must have its own temperature monitoring device such as a thermometer and/or data logger.
Immunizations refrigerator temperatures must be checked and documented twice daily when your site is open (at the beginning and end of the workday). One staff member should be assigned to this responsibility along with a back-up and all staff members should be trained in immunization storage and handling.
Temperature Monitoring Devices
Maximum-minimum thermometers provide 3 readings:
- Current temperature;
- Maximum temperature reached since the thermometer was last reset; and
- Minimum temperature since the thermometer was last reset.
Maximum-minimum thermometers must include the following features to be used for immunization monitoring:
- Be capable of reading in and out temperatures (in/out thermometer)
- Measure temperatures in increments of 0.1oC
- Be able to reset/clear the maximum and minimum readings
Healthcare providers can request a maximum-minimum thermometer at no cost from the Health Unit when ordering publicly funded immunizations. Alternatively, providers can source and purchase their own thermometers with enhanced features (e.g. with a larger display).
Guidance for setting up a maximum-minimum thermometer
- The thermometer probe should be:
- Placed inside an empty vaccine box (labelled "PROBE")
- Positioned on the middle shelf of the refrigerator
- Secured in place (i.e. with tape) to prevent from moving
- The display screen should be positioned outside of the refrigerator next to the temperature log book
- Replace batteries every 6 months
Steps for reading and recording refrigerator temperatures using a maximum-minimum:
- Write the DATE AND TIME in the vaccine temperature log book.
- Looking at the "OUT" temperatures on the thermometer displays, record the current, minimum, and maximum temperatures in the log book.
- Sign your initials beneath the recording in the log book.
- Clear/reset the thermometer history.
Call the Health Unit (519-663-5317) as soon as possible if any of the temperature recordings are outside of the recommended +2⁰C to +8⁰C range.
Data loggers are continuous temperature recording devices, which offer a historical record of refrigerator temperatures. This is particularly helpful for identifying duration if immunizations are exposed to temperatures outside of the recommended range (e.g. during a power outage).
Immunization refrigerator temperatures must still be checked and recorded twice daily when using a data logger.
- Maximum-minimum thermometers may be used in combination with data loggers for ease of monitoring and recording.
- Temperature readings from a data logger usually require computer download. Temperatures should be printed out weekly and retained for 1 year, or until your annual inspection by the Health Unit.
Immunization Transport and Temporary Storage
Cold chain must be maintained at all times, including during transportation and temporary storage.
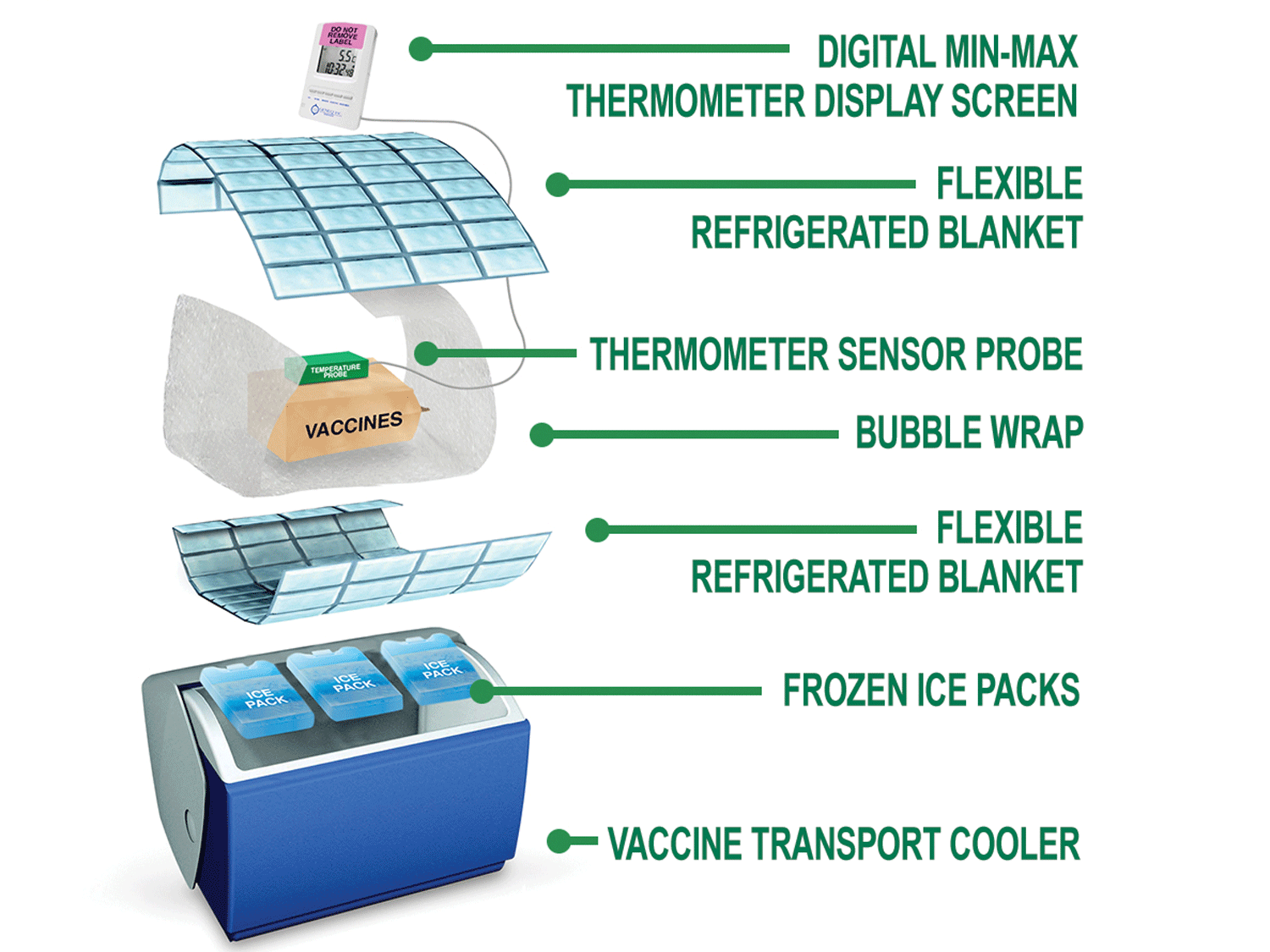
| Equipment required for immunization transport or *temporary storage: | |
|
*Temporary storage of immunizations may be necessary when cleaning a refrigerator or for emergency storage during a power outage. |
|
|
Steps for preparing and packing an insulated cooler |
|
|
An insulated cooler must be "conditioned" BEFORE it can be packed with immunizations. Conditioning involves ensuring the temperature inside the cooler is within the recommended range of 2oC and 8oC. |
|
|
Step 1
|
 |
|
Step 2
|
 |
|
Step 3
|
 |
|
Step 4
|
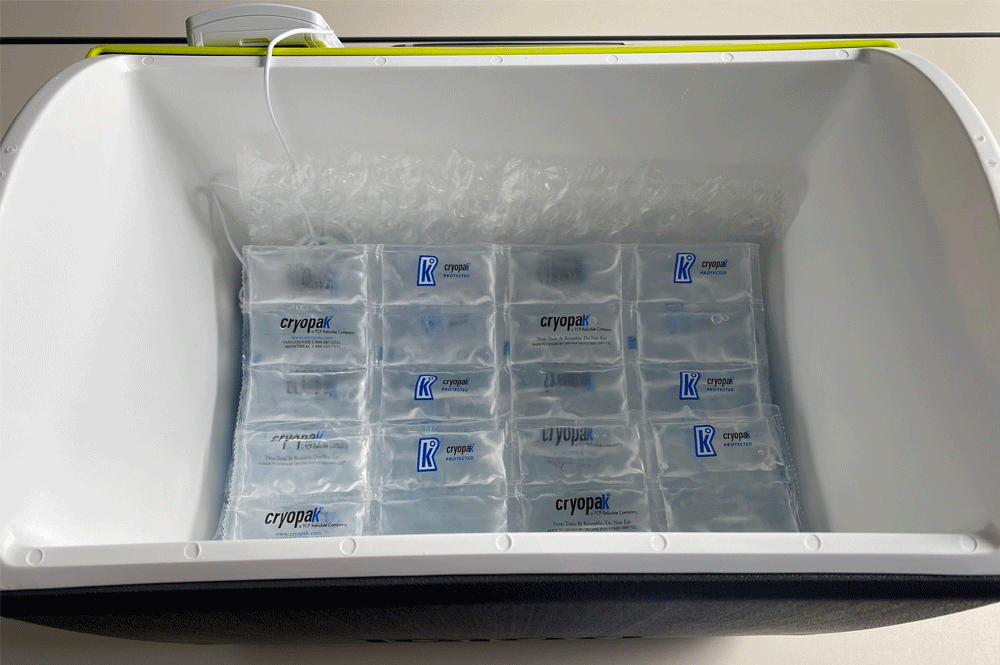 |
|
Step 5
|
 |
|
Step 6
|
 |
Temperatures of the insulated cooler should be recorded:
- when the immunizations are first packed inside the cooler,
- at least once each hour or each time the cooler is opened, whichever is more often, and
- before the immunizations are removed at the destination or being returned to the refrigerator after temporary storage.
If temperatures are not maintained between 2oC and 8oC, call the Health Unit (519-663-5317) as soon as possible and mark immunizations as DO NOT USE until it can be determined if they are suitable for administration.
Immunizations orders will not be released to couriers or office staff who do not bring appropriate packing materials and a conditioned insulated cooler to the Health Unit upon pick-up.
COVID-19 Vaccine Specifications
While there are some similarities, the storage and handling of COVID-19 vaccines requires some modifications.
COVID-19 vaccines are very fragile.
- Pfizer BioNTech Comirnaty and Moderna Spikevax COVID-19 vaccines contain mRNA surrounded by a lipid bilayer.
- This lipid bilayer is very fragile and can rupture easily with improper handling leading to wasted/ineffective vaccine.
- Take caution to ensure vials and syringes are not dropped.
- During transport:
- Label your insulated cooler as "Fragile: Handle with Care, Do Not Drop."
- Secure vials in an upright position with extra bubble wrap.
- Secure the cooler when travelling in a vehicle.
The recommended storage temperature and temperature recording guidelines for THAWED COVID-19 vaccines are the same as those listed above for other publicly funded vaccines.
See the Ministry of Health's COVID-19 Vaccine Storage and Handling resource document for additional guidance.
Publicly Funded Immunizations - Orders and Returns
The Health Unit provides publicly funded immunizations to healthcare providers, organizations and agencies who have been pre-approved and inspected.
Publicly funded immunizations that have expired or need to be wasted must be returned to the Health Unit for proper disposal.
Instructions for ordering and for returning publicly funded immunizations can be found here.
Cold Chain Incidents
A cold chain incident occurs when immunizations are exposed to temperatures outside the recommended range of 2oC to 8oC.
| Steps to take in the event of a cold chain incident |
|
Step 1
|
|
Step 2
|
|
Step 3
|
Troubleshooting when immunization refrigerator temperatures are outside of the recommended range:
- Is the temperature monitoring device is working?
- is the probe correctly placed?
- does the device need new batteries?
- are the OUT temperatures being read on the maximum-minimum thermometer?
- Is the refrigerator is plugged in?
- Has the temperature of the room changed dramatically?
- Is the refrigerator door closed/was it left opened? Is the door sealing properly?
- Is anything blocking the air flow of the refrigerator?
- If unable to determine the cause, sites may need to contact a technician to perform maintenance or repairs.
Contingency Planning
Healthcare providers, organizations and agencies should develop a contingency plan in the event of an immunization refrigerator malfunction or power outage. Contingency plans may include:
- making arrangements in advance for an alternative storage site (e.g. another office/pharmacy)
- using insulated containers and packing materials
- in the event of an outage where power is expected to be restored within several hours - add ice packs to the refrigerator and keep the door closed
Temperatures should be monitored and recorded while your contingency plan is in effect. Call the Health Unit (519-663-5317) as soon as possible if the immunizations are exposed to temperatures outside of the recommended range.
Last modified on: August 18, 2025
References
https://files.ontario.ca/moh-vaccine-storage-handling-protocol-en-2018.pdf
https://files.ontario.ca/moh-ophs-ref-vaccine-storage-handling-guidelines-2021-en.pdf
https://www.ontario.ca/files/2024-09/moh-covid-19-guidance-vaccine-storage-handling-en-2024-09-26.pdf